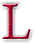
ike all the chapters in this section,
we start with an immensely simple idea. In this case, the
simple observation (and fundamental principle in economics)
that "bad money drives out good." Expanded to assume
a more interdisciplinary utility, we might say that when
you dilute "the good" in any system by allowing the
introduction of "the bad," you succeed in getting rid of
the good altogether, in varying degrees and in proportion
to the bad you allow to flourish.

A good example of this
might be the internet. Email has become a vital tool of
business and industry -- not to mention what it has
done for personal communications. In the early days of
the internet, I found it to be an enormous benefit.
Then, over time, we saw the introduction of more and more
"spam," to the point where many users feel overloaded in
attempting to "extract the good messages." I myself got to the
point where even with advanced filtering methods, I was
overwhelmed and urging my associates to CALL ME on the phone
if their message was IMPORTANT. I could not take the chance
that an important message might be missed awash all the
unsolicited garbage.

No one likes spam.
And the laws to try to reduce or eliminate its overgrowth
in cyberspace do not appear to be working. There are, however,
far more sinister applications of Gresham's Law, involving
far more deliberate attempts to drive out the good. And despite
arguments that the converse of Gresham's Law holds
historical accuracy, there is no evidence that the
role of organized medicine -- at any of its levels
of operation -- has served to use the good to drive out
the bad.
1.

Because I am
an herbalist, people are sometimes shocked to hear me
say that at least 90% of the herbal products on the
market are, at best, marginal. Most products are,
to use the vernacular, just garbage.

Herbology,
well practiced with an eye towards maximizing the
desired effect on the customer, is an exacting
science. Depending on the herb and its use,
and in varying degrees, the efficacy of a given herbal
product will be based on when it is picked, how
it is processed, the proven efficacy of the
mixed ingredient formula itself (where applicable),
and the validity of the protocol provided, etc.

Good
manufacturing practices should be enforced to
ensure the very best herbal products are sold
on the market. But such standards are not
used OR enforced, and the reason is simple:
the FDA doesn't recognize herbal supplements as
anything more than that: a dietary supplement.
Herbal manufacturers get to hide behind the DSHEA
disclaimer -- after all, if you tell your customer
up front, "This product is not designed to
diagnose, prevent, treat or cure any disease,"
then if you market a medicinally worthless product
how can you be held accountable?

You can't.


Officials at
the FDA know this. They know that the marketplace
is awash with worthless herbal products that do not
benefit the customer. They like it that way.
The pharmaceutical companies like it that way.
And most physicians like it that way. By taking
a blind eye to having near non-existent standards
for medicinal herb products you have in place a
phenomenon where bad products drive out the good.
In other words, consumers stay confused and distrustful
of good herbalism in general.

For every
customer who has tried a product like Cansema and
had a spectacular result, there are 20 who honestly
believe that herbs don't work. And why shouldn't
they believe this? They bought some cheap $4.00
bottle of product from WalMart, took it home,
tried it, got no result, so what conclusion should
they draw?

Another good example
of this phenomenon is the regulatory posture towards
homeopathic preparations. Although the FDA takes a
conciliatory position towards homeopathy
on
its web site.
2, its lack of
enforcement of good manufacturing practices has lead
to an unusual chasm in how homeopathy is perceived
in the U.S. versus other countries that are more open
to alternative health care practices.

A close associate
of mine in New York is a native of India who came
to the U.S. 20 years ago to get his MBA. He has
indicated to me that he knows physicians in
Mumbai (Bombay) who use homeopathic preparations
regularly, along with ayurvedic, herbal, and
more conventional drug therapies. He has told me,
"I didn't realize why Americans didn't think more
highly of homeopathic remedies until I came to
understand that because of unenforced standards
your homeopathics don't work as well -- if at all."

Now, I have
to regard this commentary as hearsay at this point.
In all our work at Alpha Omega Labs, despite the
fact that we manufactured, in part, most of the
350 different products we carried, we never
manufactured any homeopathics. So this is not my area
of specialty.

Nonetheless,
given my experience in other areas where Gresham's Law
runs amok, I regard this as another example of
a phenomenon where the FDA, in allegiance to the
pharmaceutical industry, turns a blind eye to
encouraging the best alternatives to flourish in
the market -- simply because by allowing the bad
to dilute the public's confidence in the good,
consumers are left only with the choice of
going with more expensive pharmacological
therapeutic options.
- http://www.columbia.edu/~ram15/grash.html
The twelve and final section ("Conclusions") of
this Columbia University essay attempts to make just
such an assertion. To-wit: "The catchy phrase, 'bad money drives out good,' is not a correct statement of Gresham's Law nor is it a correct empirical assertion. Throughout history, the opposite has been the case. The laws of competition and efficiency ensure that 'good money drives out bad.' The great international currencies--shekels, darics, drachmas, staters, solidi, dinars, ducats, deniers, livres, pounds, dollars--have always been "good" not "bad" money."
- http://www.fda.gov/fdac/features/096_home.html
This piece was abstracted from the December, 1996, issue of FDA Consumer magazine.





